Researchers conducted experiments on carbon-based materials and were able to develop an outstanding “glass” with peculiar features and various potential applications, as stated in the Independent online newspaper and corroborated by National Science Review, where the original research paper can be fully accessed.
We found this subject so interesting that we decided to share it with you.
This article contains the following topics:
- INTRO
- RESULTS AND DISCUSSION
- Structural characterisation
- Mechanical properties
- Optical properties
- Comparison of various types of AM carbon materials or “Glass”-state materials
- METHODS
- CONCLUSION
1. INTRO
Exploring new forms of carbon has always been a matter of interest amongst scientists. In 2021, researchers conducted experiments with amorphous carbon materials (AM-I, AM-II and AM-III) and presented their findings.
The results of comprehensive mechanical tests performed in the synthesised amorphous carbon material named AM-III, prove that this is the hardest and strongest AM material know to this day – it can almost reach diamond crystal’s strength.
AM-III demonstrates outstanding mechanical and electronic properties, ultrahigh strength and wear resistance. A material with these characteristics, can be potentially used in photovoltaic applications, like solar cells or other technological applications, due to its properties.
This research is a demonstration of a “systematic study of the behaviour of C60 fullerene at the previously unexplored pressure of 25 GPa and different temperatures”.
The materials named AM-I, AM-II and AM-III were synthesised and characterised by the following techniques:
- X-Ray diffraction (XRD)
- Raman spectroscopy
- High-Resolution Transmission Electron Microscopy (HRTEM)
- Electron Energy Loss Spectroscopy (EELS)
As said in the original paper:

Once the AM-III was the most interesting of the three AM materials, we’re going to focus on this one on the next sections of this article.
2. RESULTS AND DISCUSSION
Structural characterisation
After the treatment of C60 at 25 GPa and an increase in temperature (from 1000°C to 1200°C) the material changes colour from opaque black to transparent yellow, as we can see in the image bellow (picture C):
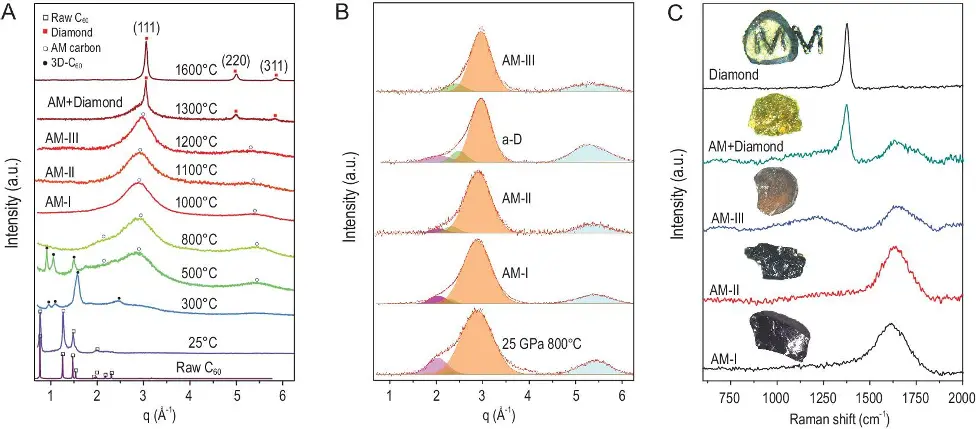
Image Source: NSR
Mechanical properties
- AM-III has the highest hardness of all known AM materials;
- The hardness can rival that of diamond;
- Measurements demonstrate that the AM-III is comparable in strength to diamond and superior to the other known high-strength materials;
- A set of experimental and theoretical results demonstrate that ultrahigh hardness and strength comparable to crystalline diamond can be achieved.
Optical properties
Quoting the original piece:
“In view of the yellow-transparent nature of AM-III, its visible light absorption spectrum was measured in transmission utilising a diamond anvil cell (DAC).”
These AM carbon materials are a class of semiconductors with bandgaps less than diamond and close to the AM silicon films, often used in many ways in the technological industry.
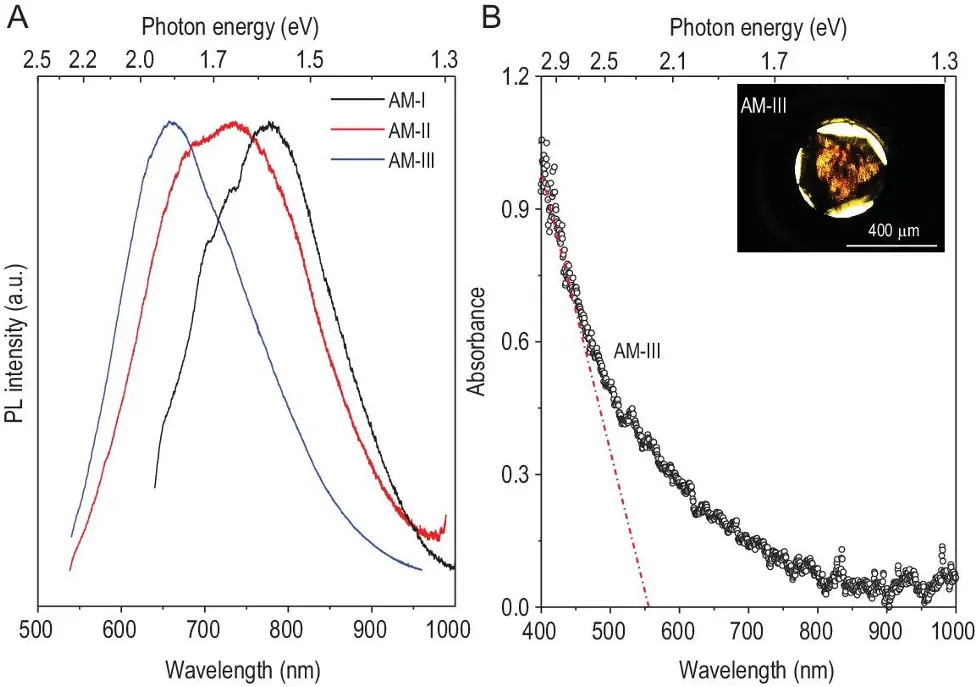
Image Source: NSR
Comparison of various types of AM carbon materials or “Glass”-state materials
When testing crystalline materials, using one technique alone (XDR, for example) is enough to analyse its structure, whereas AM carbon materials need complimentary characterisation for a clearer identification of the different states of disordered matter.
A unique combination of semiconductive and superior mechanical properties – hardness and strength at the level of natural/synthetic diamond – is provided by a distinct short-range order, microstructure and composition.
3. METHODS
For this experiment were performed several tests:
- Sample synthesis;
- X-ray diffraction and Raman spectroscopy;
- HRTEM and EELS measurements;
- Hardness and elastic modulus measurement;
- Compressive strength test;
- Optical absorption;
- Thermal stability measurement.
For more information on these methods, make sure to read the original research article.
4. CONCLUSION
In conclusion, AM carbon materials — AM-III in particular — show outstanding results concerning mechanical properties, hardness and strength.
So far, we know that these materials are ultra-hard, ultra-strong, semiconducting and with various purposes, like photovoltaic or technological applications. Still, it cares for further theoretical and experimental investigation to better understand its composition and other potential applications.
If you find this article interesting, make sure to leave a comment with your thoughts and we’ll reply ASAP. In the meanwhile, you can read other articles from our blog.







